 
Multiple Failure Modes Analysis and Weighted Risk Priority Number Evaluation in FMEA. Xiao, Ningcong., Hong-Zhong, Huang., Yangfeng, Li., Liping, He. 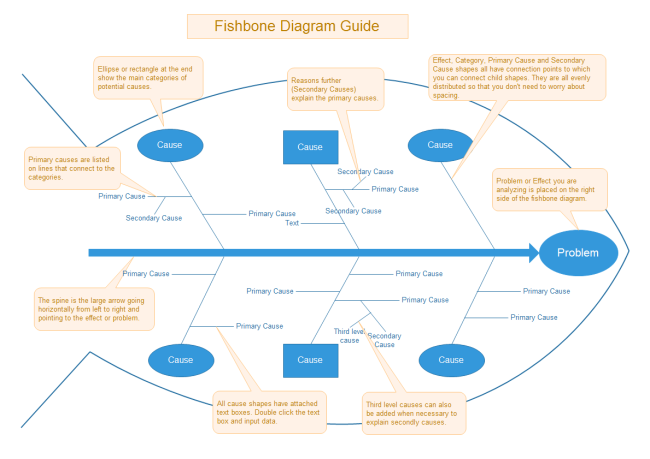
Using an Ishikawa Diagram as A Tool to Assist Memory and Retrieval of Relevant Medical Cases from the Medical Literature. Management System in Production Engineering. Problem solving techniques as a part of implementation of six sigma methodology in tire production – Case Study. WHO Guideline on Quality Risk Management. Combining FMEA with DEMATEL Models to Solve Production Process Problems. Tsai, Sang-Bing., Jie, Zhou., Yang, Gao., Jiangtao, Wang., Guodong, Li., Yuxiang, Zheng., Peng, Ren. International Journal of Computer Integrated Manufacturing. An Evaluation of Failure Modes and Effect Analysis Generation Method for Concepttual Design. International Journal Health Care Quality Assurrance. Evaluating Inputs of Failure Modes and Effects Analysis in Identifying Patient Safety Risks. Failure Mode and Effects Analysis Output : Are They Valid?. Journal of Textile Science and Engineering. Use of Quality Tools for Problem Analysis (FMEA and Ishikawa Diagram) in a Small Textile Business. International Research Journal of Engineering and Technology. Quality Improvement Using FMEA : A Short Review. International Journal of Recent Technology and Engineering. Quality Improvement of Capacitors through Fishbone and Pareto Techniques. International Journal of Scientific & Engineering Research. Application of Pareto principle and Fishbone diagram for Waste Management in a Powder Filling Process. IOP Conference Series : Materials Science and Engineering. A New Model of Ishikawa Diagram for Quality Assessment. Bulletin of Faculty of Pharmacy Cairo University. Quality risk management during pharmaceutical ‘good distribution practices’ – A plausible solution. International Journal of Research in Pharmacy and Chemistry. A Review on Failure Mode and Effect Analysis – A Tool of Quality Risk Management Based on ICH 9. Application Of Fishbone Diagram To Determine The Risk Of An Event With Multiple Causes. Importance of Quality Risk Management Pharmaceutical Quality System : Recent Trends and Harmonization. A Beginner’s Guide to Quality Risk Management. International Journal of Pharmaceutical Quality Assurance. Quality Risk Management (QRM) in Pharmaceutical Industry : Tool and Methodolgy. 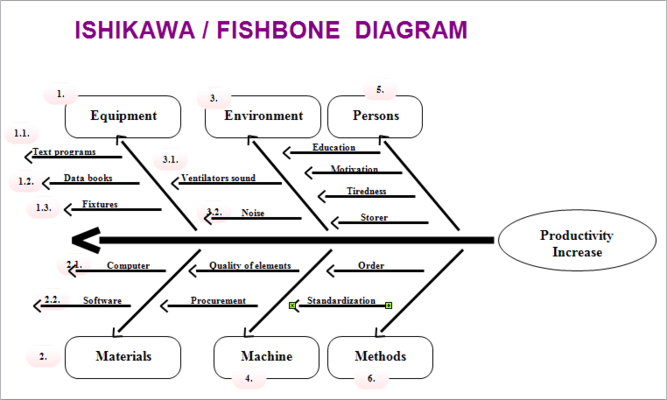
International Journal of Product Lifecycle Management. Decision-making in Product Quality Based on Failure Knowledge. A Modified Failure Mode and Effect Analysis Method for Supplier Selection Problems in The Supply Selection Problems in The Supply Chain Risk Environment: A Case Study. Application of Fishbone Analysis for Evaluating Supply Chain and Business Process- A CASE STUDY ON THE ST JAMES HOSPITAL International of Managing Value and Supply Chain. PerKa Badan Pengawas Obat dan Makanan Republik Indonesia No. Dapat disimpulkan dengan menggunakan metode dalam manajemen risiko mutu dapat mengendalikan risiko secara efisien.īadan Pengawas Obat dan Makanan. Hasil dari menggunakan kedua metode adalah dapat memfasilitasi keputusan yang lebih baik, memberikan jaminan yang lebih besar untuk menghadapi risiko potensial, serta dapat memengaruhi tingkat pengawasan proses produksi maupun produk akhir. Tujuan dari penulisan ini adalah untuk menelaah kedua metode tersebut dalam manajemen resiko sehingga dapat digunakan secara proaktif dan reaktif. Penulisan ini berdasarkan studi literatur melalui buku dan jurnal penelitian yang telah diterbitkan dari 2009 hingga sekarang dan dapat diakses secara online di website jurnal nasional dan internasional mengenai Ishikawa Diagram dan FMEA sebagai metode untuk manajemen risiko. Metode yang sering digunakan dalam manajemen risiko adalah Ishikawa Diagram dan Failure Mode Effect Analysis (FMEA) karena dapat memberikan banyak informasi mengenai penyebab masalah dan akibatnya serta mudah digunakan. Manajemen risiko yang efektif yaitu dapat memastikan kualitas produk terjamin secara proaktif dan reaktif selama pengembangan, proses produksi, hingga beredarnya produk di pasaran. Salah satu metode untuk menjaga kualitas produk yaitu dengan manajemen risiko mutu. Kualitas produk harus dijaga sepanjang siklus hidup produk sehingga kualitasnya tetap konsisten dan aman untuk digunakan. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
